Detecting Sealing Defects in Medicine Bottles via Infrared Monitoring
Challenge
Ensuring uniform and complete induction sealing in pharmaceutical bottles is difficult due to the rapid production pace, small measurement area, and lack of real-time visual confirmation, making early detection of sealing defects challenging and increasing the risk of contamination, product loss, and costly recalls.
Solution
Real-time thermal monitoring of the cap area immediately after induction sealing enables early detection of incomplete or faulty seals. By capturing localized temperature deviations on moving bottles, the system ensures defective units are identified and rejected before capping, without interrupting the production flow.
Benefits
- Detects sealing defects instantly, preventing faulty products from continuing down the line
- Reduces product waste and rework by identifying issues early in the process
- Enhances safety by ensuring consistent sealing and preventing potential contamination
- Maintains high production speeds without compromising quality control
- Supports process optimization through stored thermal data and event-triggered analysis
Challenges in Detecting Sealing Defects Before Cap Application
The sealing of medical bottles is a critical step in pharmaceutical manufacturing, as it ensures the integrity and safety of the product. This process involves placing an aluminum foil seal beneath the plastic bottle cap, which is heated and melted using induction sealing technology. The primary challenge in this process is ensuring the seal is complete and uniform. Any defects in the seal, such as gaps, incomplete melting, or excessive heat leading to damage, can result in compromised bottle integrity. Such defects may cause product contamination, leakage, or degradation, leading to product recalls or safety concerns.
The induction sealing process itself is intricate. It involves multiple layers: a wax layer, an aluminum foil, and pulp backing, all of which must be heated precisely for the seal to form correctly. The induction machine generates heat through electromagnetic fields, which causes the wax to melt, bonding the aluminum foil to the bottle opening. However, due to the rapid nature of the production line, identifying sealing defects before the bottle reaches the final capping stage is difficult. There is no visual confirmation of a proper seal at the moment of sealing, and any issues are typically only noticed later in the production cycle, when it is often too late to correct the defect without disrupting the workflow.
The primary challenge we face is the real-time detection of these defects, which is crucial for immediate corrective action. The fast pace of pharmaceutical production demands a system that can monitor each bottle accurately without slowing down the line. Furthermore, the sealing process involves a small area on the bottle, making it critical to have precise and localized temperature measurements. Inaccurate or delayed detection of sealing issues can lead to significant financial losses due to product waste and can jeopardize the safety and efficacy of the product. Your role in this process is vital, and your expertise is needed to ensure the quality and safety of our products.


Automating Defective Bottle Rejection by Monitoring Cap Sealing Temperature Deviations
To address the challenges of detecting improper sealing in medical bottles, the pharmaceutical company Momenta, in partnership with distributor Contemp, implements Optris advanced thermal imaging technology. The Optris Xi 400 infrared camera is chosen because it delivers high-speed, real-time thermal measurements, making it ideal for fast-paced pharmaceutical production environments.
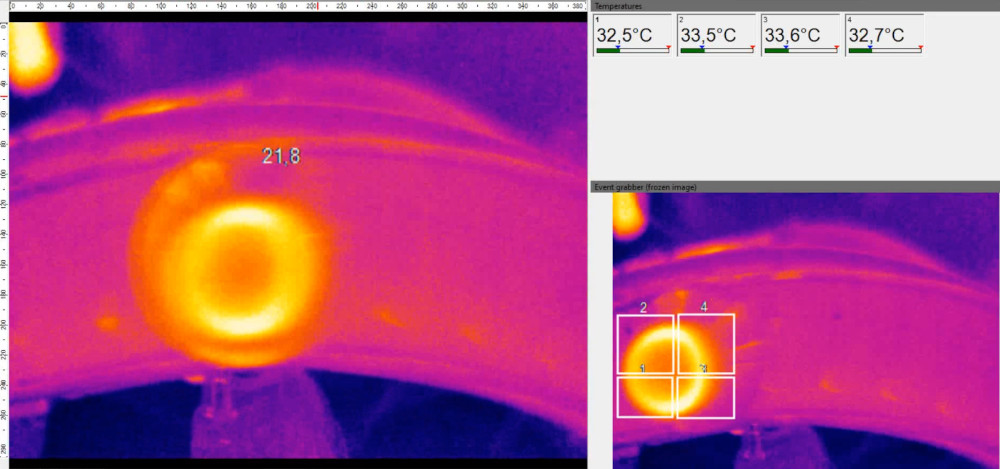
The Xi 400 camera is positioned above the induction sealing machine, where it monitors the heat transfer from the aluminum foil to the plastic bottle cap. The induction machine melts the wax layer between the foil and the cap to create a proper seal. Capturing the surface temperature of the cap immediately after the induction process allows the camera to detect any inconsistencies in sealing, which is crucial for ensuring product safety.
The Xi 400’s 80Hz frame rate is critical, enabling the system to capture rapid temperature changes as bottles move through the sealing station. This high sampling frequency prevents motion blur, providing clear, precise thermal images for detecting faulty seals. The camera analyzes multiple zones on the cap surface to ensure the seal is complete.
Integrated with the Optris PI NetBox, the Xi 400 system eliminates the need for an external computer, simplifying installation. The temperature data is processed using PIX Connect software and fed into the automation system. If the temperature in any monitored zone falls below the alarm threshold, the digital output triggers an alarm and activates a rejection mechanism that removes defective bottles from the production line.
The Event Grabber function within PIX Connect captures thermal images at critical moments, allowing operators to review and analyze the sealing process in detail. This feature provides valuable snapshot analysis and supports quality assurance by storing images for further evaluation. Additionally, color palettes in the software make it easy for operators to identify temperature inconsistencies, enhancing the system’s visual feedback.
Easy Integration and Operation with Minimal External Equipment
The implementation of the Optris Xi 400 infrared camera system brings significant results and benefits to the pharmaceutical company Momenta, enhancing both product quality and operational efficiency. One of the primary benefits is the ability to detect improper sealing in real-time. By monitoring the surface temperature of the plastic cap after the induction process, the system identifies defective seals immediately, ensuring that only properly sealed bottles continue down the production line. This real-time detection helps reduce the risk of product contamination or spoilage, a critical factor in maintaining the safety and efficacy of pharmaceutical products.
The high frame rate and precision of the Xi 400 camera allow it to capture even the smallest temperature variations on the cap surface, offering comprehensive quality control. With four distinct zones monitored for temperature consistency, the system can detect whether any part of the cap has not sealed correctly. Bottles with temperature readings below the defined threshold are automatically rejected from the production line, preventing defective products from reaching consumers and minimizing the potential for costly recalls.
Additionally, the integration of the Event Grabber function within PIX Connect provides operators with the ability to store thermal images for future analysis, ensuring thorough quality assurance. This feature also supports process optimization by allowing operators to review past thermal data, helping identify upstream issues such as inconsistencies in the wax layer or problems with the aluminum foil. By pinpointing the root cause of any sealing defect, the company can take proactive steps to refine the process, improving overall production efficiency.
The Optris solution not only ensures a reliable and automated method of detecting sealing issues but also reduces waste and rework by catching defects early. Its ease of use, combined with the system’s fast response and robust automation integration, positions Optris as the preferred partner for ensuring quality control in pharmaceutical bottle sealing processes.
Recommended Products
Other Pharmaceutical & Medical Applications

Talk to us about your IR Temperature Measurement Requirements
There are over 300 different pyrometer variants to choose from in the Optris infrared pyrometer portfolio each optimized for material, spot size, distance from the target, and environmental conditions. Fortunately, there is a trained engineer to phone or chat with to guide you through the process of choosing the perfect infrared sensor for your application.
The same support is available for the extensive IR camera product line.