Kirchhoff’s Law of Thermal Radiation
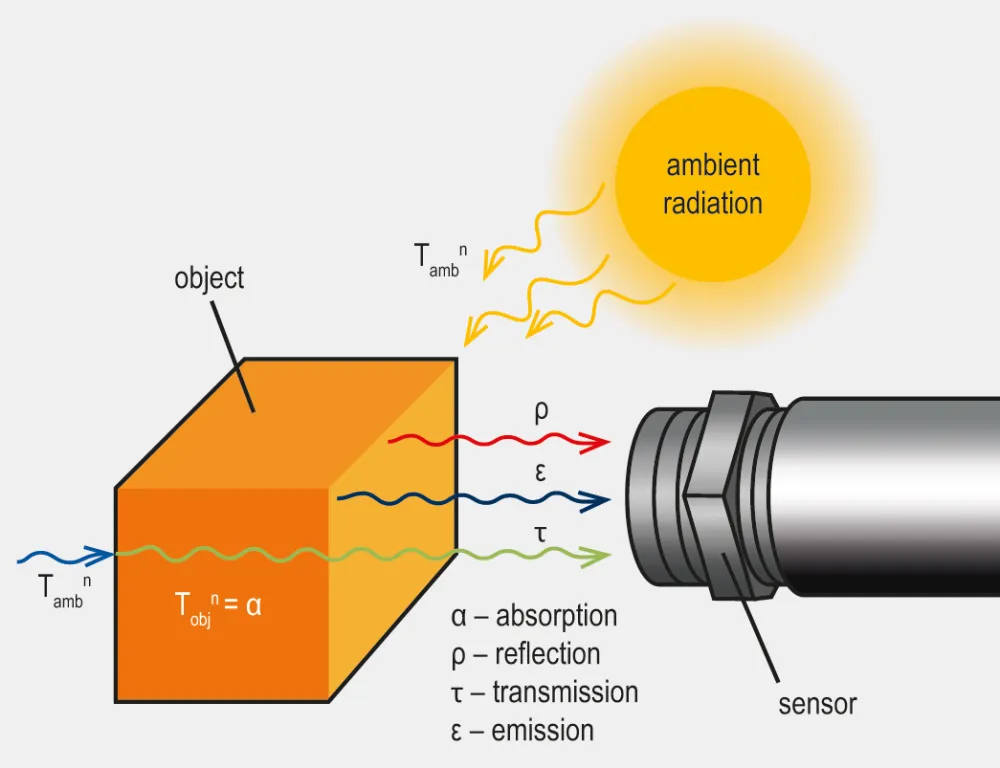
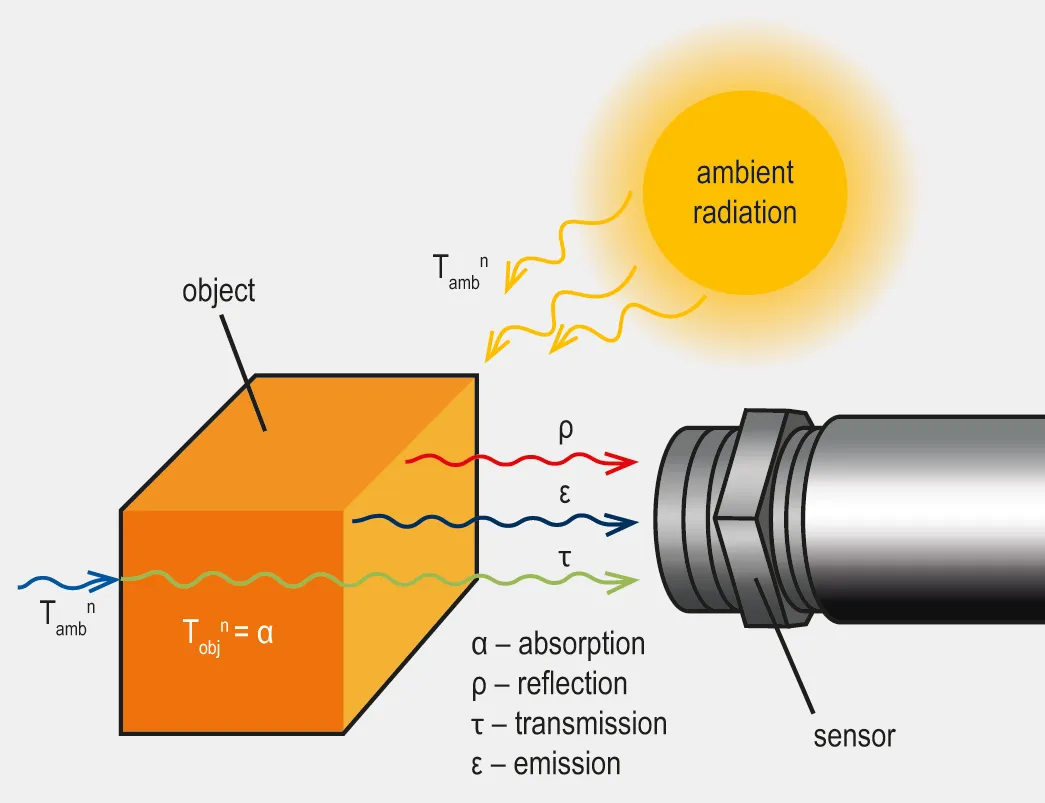
In the realm of infrared radiation, Kirchhoff’s Law states that a material in thermal equilibrium has an emissivity equal to its absorptivity at a specific wavelength.
Emissivity refers to how effectively a material emits infrared radiation in comparison to a perfect blackbody at the same temperature, while absorptivity measures how effectively a material absorbs incoming infrared radiation. A material with high absorptivity can absorb most of the radiation it receives, while a material with low absorptivity reflects or transmits more radiation. A blackbody has an emissivity of 1, meaning it emits the maximum possible radiation at a given temperature and wavelength, whereas real-world materials have emissivities of less than 1.
Kirchhoff’s Law is applicable to materials in thermal equilibrium, meaning their temperature remains constant without gaining or losing heat. Under these conditions, the energy absorbed by the material is equal to the energy it emits. This equilibrium is vital, as the equality of emissivity and absorptivity generally holds true only when the material is in thermodynamic equilibrium.
Another implication of Kirchhoff’s Law is that emissivity cannot exceed one, as absorptivity is also constrained by the conservation of energy. Consequently, a material cannot radiate more thermal energy than a blackbody at equilibrium.
Back to LexiconRecommended Products

Talk to us about your IR Temperature Measurement Requirements
Our Infrared Temperature Measurement experts can help you find the right Optris product for your application.