Planck’s Law
Planck’s Law describes the electromagnetic radiation emitted by a blackbody in thermal equilibrium at a given temperature. Its application extends from fundamental physics to practical technologies such as infrared thermometry, enabling precise non-contact temperature measurements across various industries. Planck’s Law explains how objects emit radiation and provides the theoretical basis for many modern scientific and industrial processes.
Formulated by Max Planck in 1900, this law provided a pivotal understanding of quantum mechanics and radiation physics. It explains how the intensity of radiation varies with wavelength and temperature, revolutionizing our comprehension of heat and light emission.
A perfect blackbody, a theoretical construct, is an idealized physical body that absorbs all incident electromagnetic radiation, regardless of frequency or angle of incidence. It then emits this energy perfectly, with the spectrum of the emitted radiation solely dependent on the body’s temperature. Real-world objects, while not perfect blackbodies, can approximate their behavior to varying degrees.
Planck’s Law quantifies the spectral radiance of a blackbody, describing the amount of energy emitted per unit area, per unit time, per unit solid angle, and per unit wavelength:
[math]B(\lambda,T) = \frac{2hc^{2}}{\lambda^{5}}\frac{1}{e^{\frac{hc}{\lambda kT}}-1}[/math]
where:
• [math]B(\lambda,T)[/math] is the spectral radiance.
• [math]\lambda[/math] is the wavelength.
• [math]T [/math]is the absolute temperature of the blackbody.
• [math]h [/math] is Planck’s constant ([math]6,626069\cdot10^{− 34}J ⋅ s[/math]).
• [math]c [/math] is the speed of light in a vacuum ([math]\cong3\cdot10^{8}\frac{m}{s}[/math]).
• [math]k [/math] is the Boltzmann constant ([math]1,380649\cdot10^{-23}\frac{J}{K}[/math])
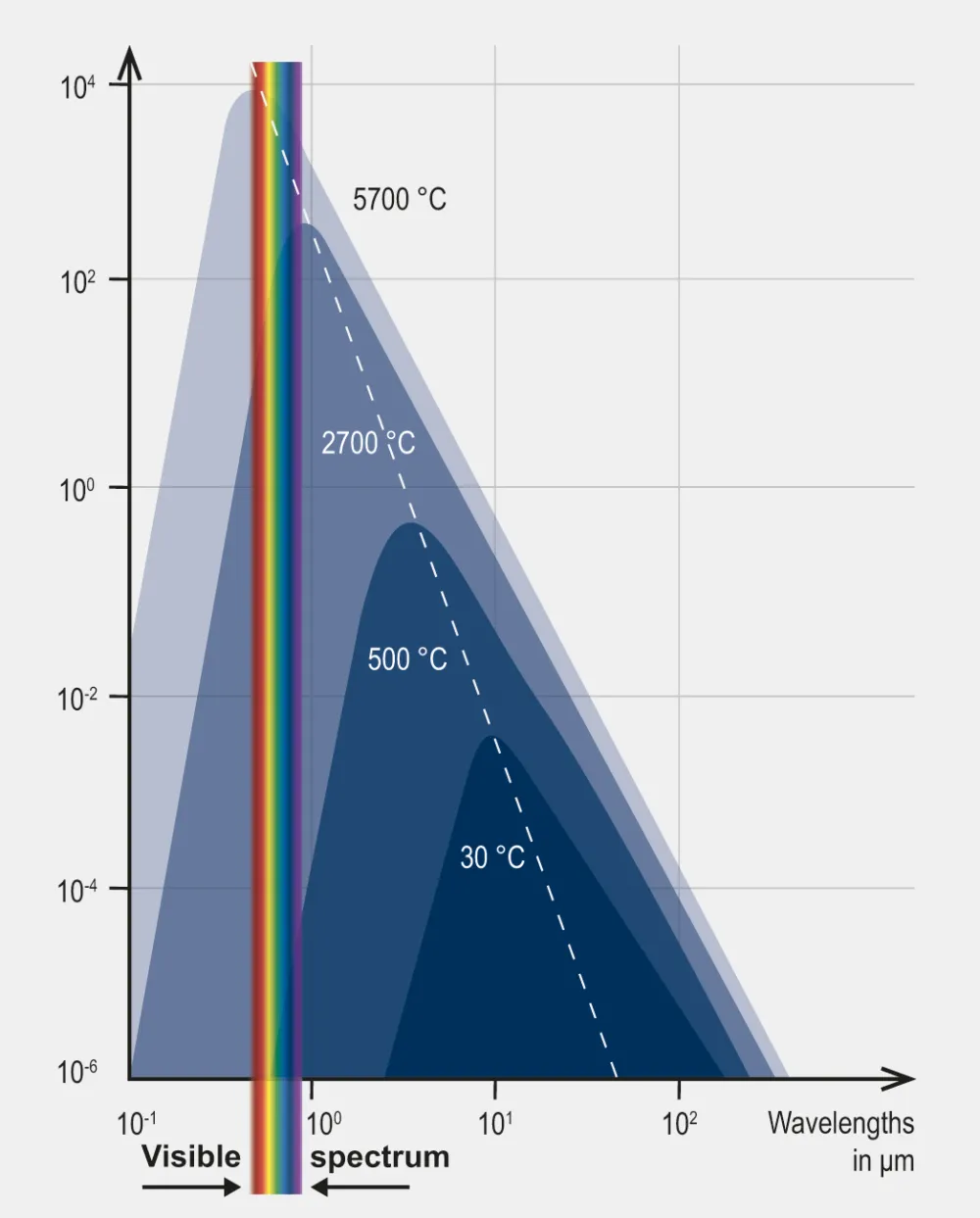
Planck’s Law shows that the radiation intensity increases with temperature and shifts to shorter wavelengths as the temperature rises. This is why hotter objects emit more radiation and appear brighter and bluer. The law also indicates that no radiation is emitted at very short wavelengths (high frequencies) and that the intensity at longer wavelengths (low frequencies) increases with temperature but at a diminishing rate.
Planck’s Law leads to two important derived laws Wien’s Displacement Law and Stefan-Boltzmann Law. Wien’s Displacement Law states that the wavelength at which the radiation from a blackbody is at its maximum (peak wavelength) is inversely proportional to the temperature. Stefan-Boltzmann Law states that the total energy radiated per unit surface area of a blackbody per unit time (the blackbody’s radiant exitance) is directly proportional to the fourth power of the blackbody’s temperature.
Back to LexiconRecommended Products

Talk to us about your IR Temperature Measurement Requirements
Our Infrared Temperature Measurement experts can help you find the right Optris product for your application.